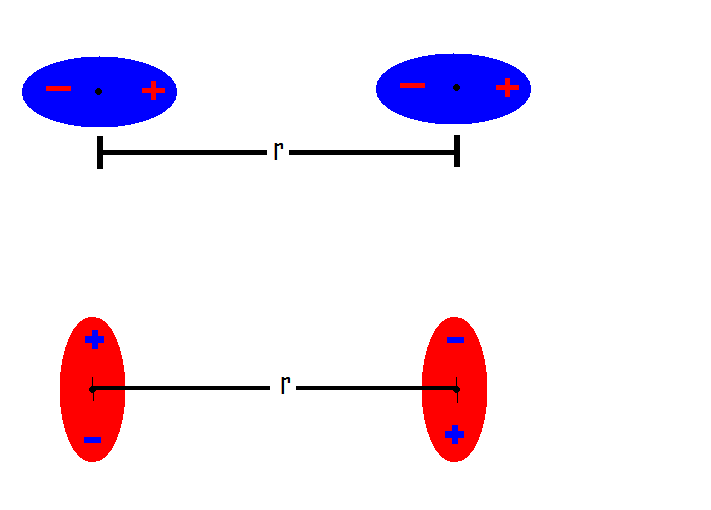
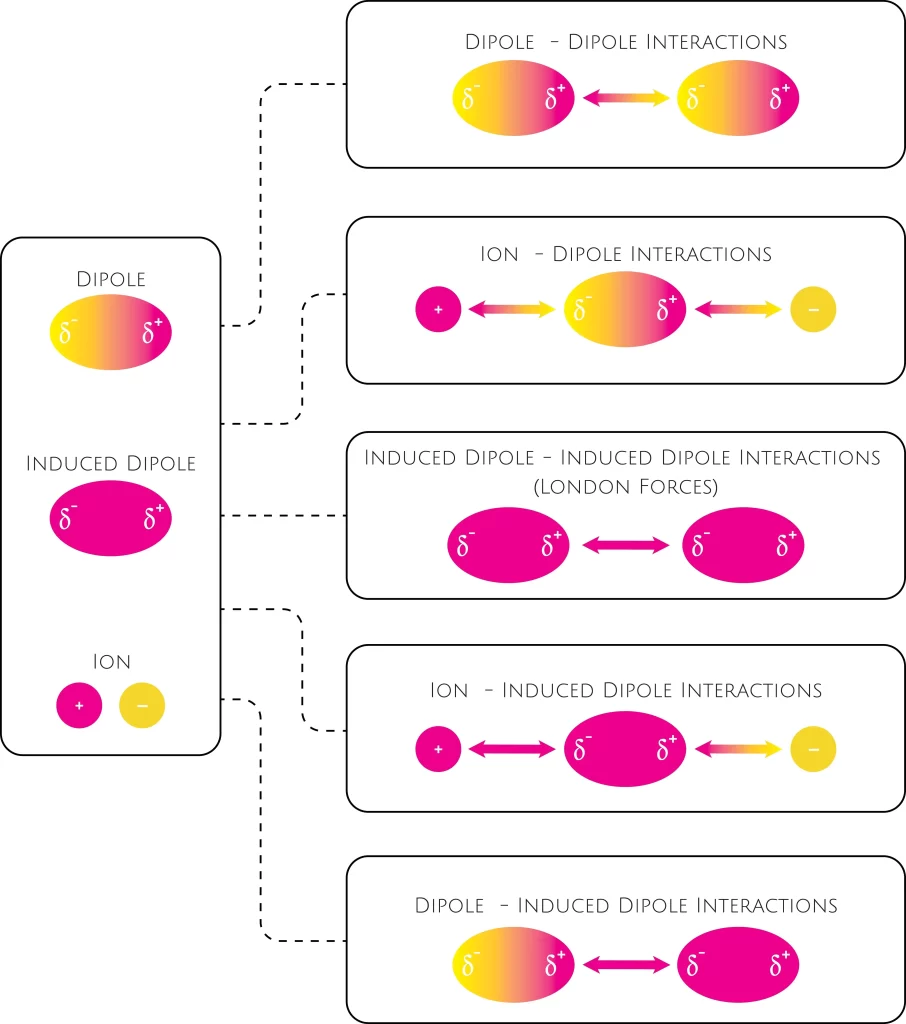
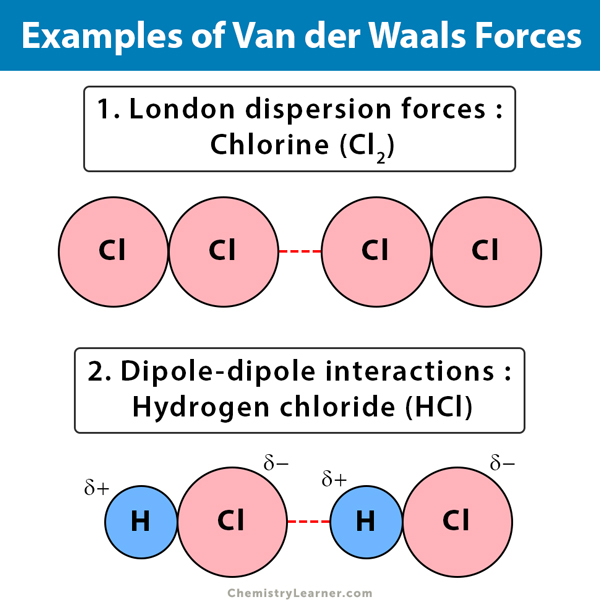
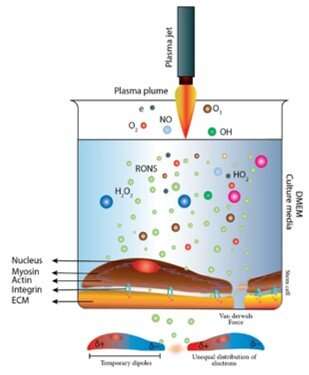
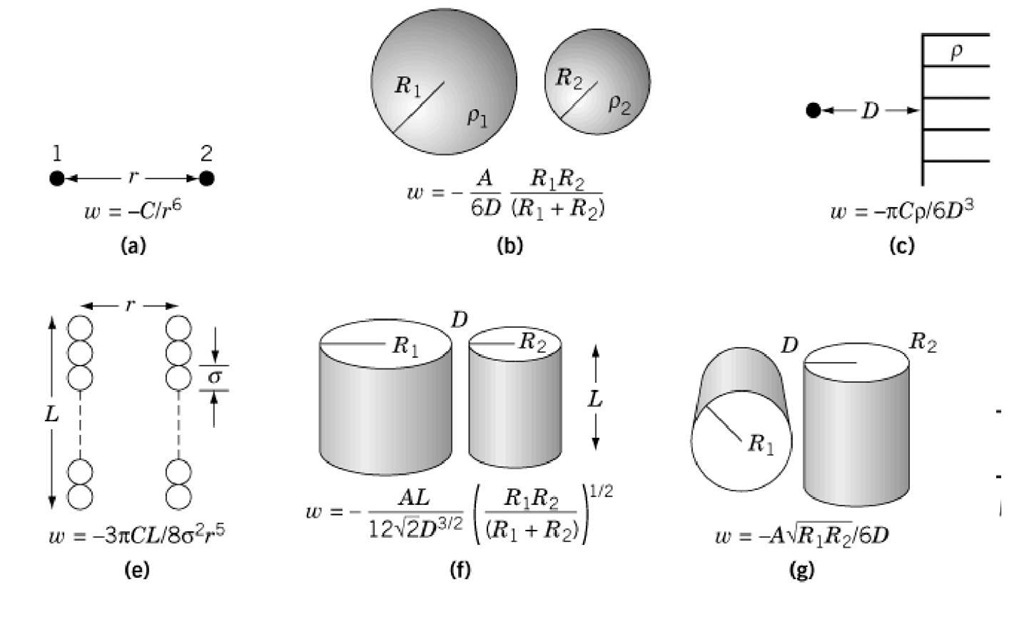
Novel analytical expressions for determining van der Waals interaction between a particle and air–water interface: Unexpected stronger van der Waals force than capillary force - ScienceDirect

Les interactions de Van Der Waals ( Les interactions intermoléculaires ) @worldofbiology3216 - YouTube
Van der waals force is a distance-dependent interaction between atoms or molecules. Unlike ionic or covalent bonds.the weak dipole attraction . physics and chemistry concept. 28282119 Vector Art at Vecteezy