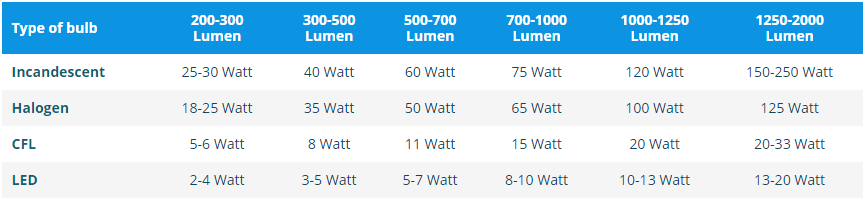
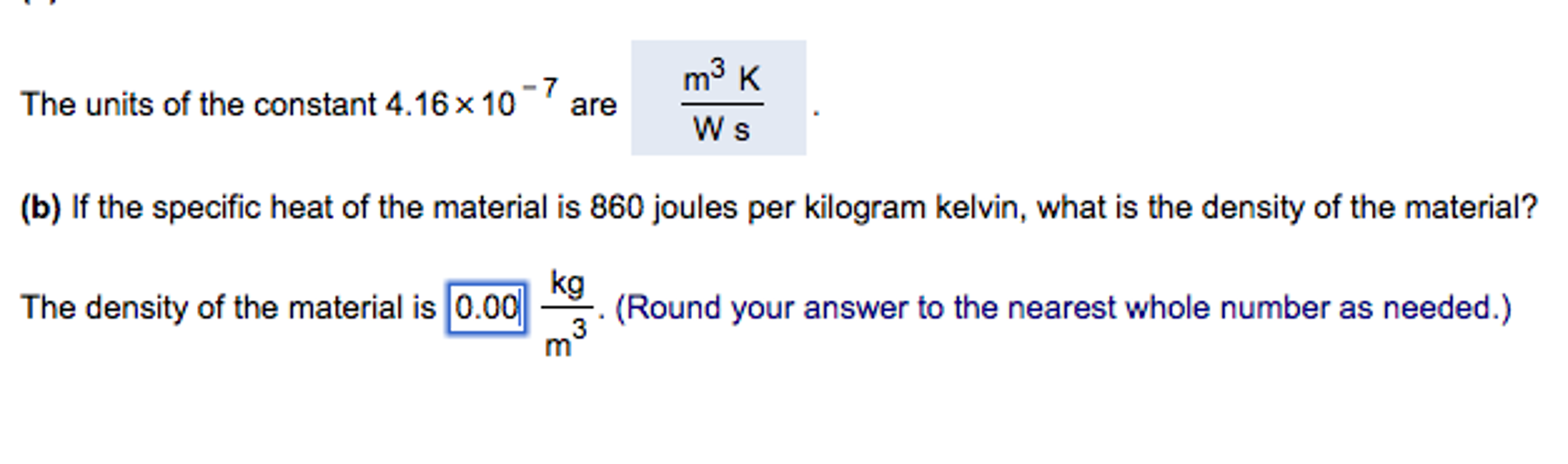
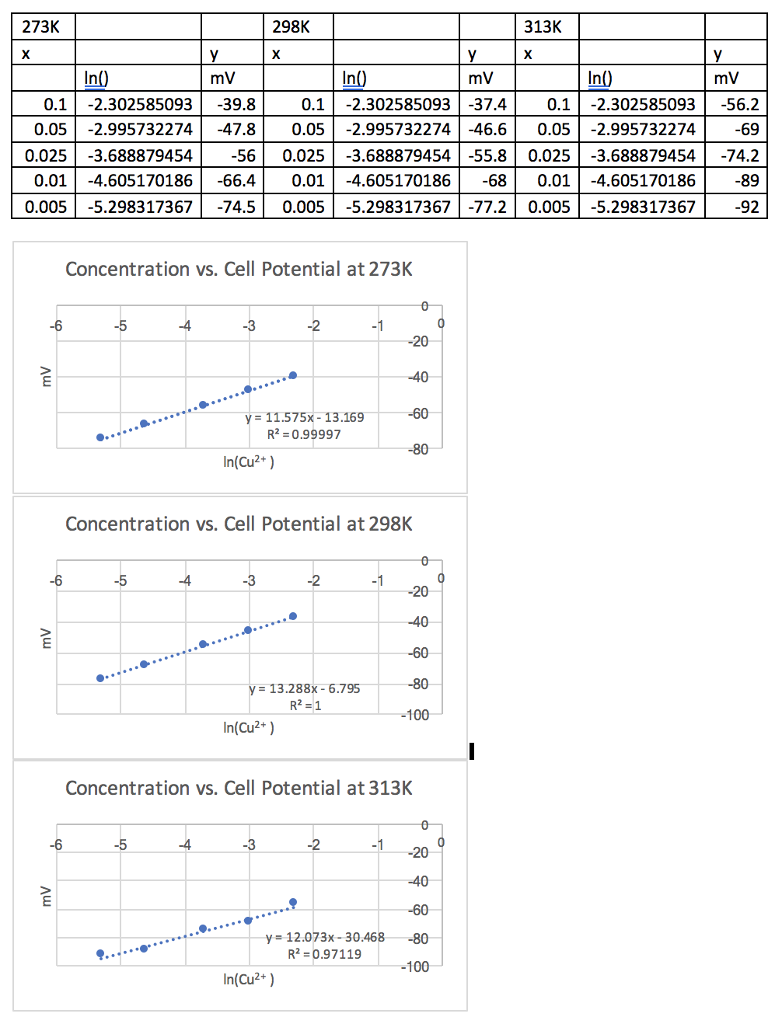
Specific heat of hydrogen at constant pressure is 30 joule per Kelvin per mol. If unit of length changed to 50 cm, unit of time changes to 1/4 sec and unit of

SOLVED: Express your answer in joules per kelvin to three significant figures. Azd 10^23 J/K = 4.201 J/K Submit Previous Answers Request Answer Incorrect; Try Again; 4 attempts remaining Part C Complete

SOLVED: The Ideal Gas Constant has units of Joule * (mole)-1 (Kelvin)-1. In SI base units this is: kg m2 s-2 K-1 mol-1 kg m K-1 mol-1 s-1 kg m2 S K-1

The quantity of energy as heat that must be transferred to raise the temperature of 1 gram (1g) of a substance 1 Kelvin (1K) Specific Heat Definition. - ppt download

The enthalpies of formation of n2o and no at 298 Kelvin and 82 and 90 kilo joule per mole the enthalpy of the reaction n2o gas half O2 gives to 2no? - EduRev Class 11 Question
4 gram of a gas occupies 22.4 litres at NTP the specific heat capacity of the gas at constant volume is 5 joule per Kelvin per mole. If the speed of sound