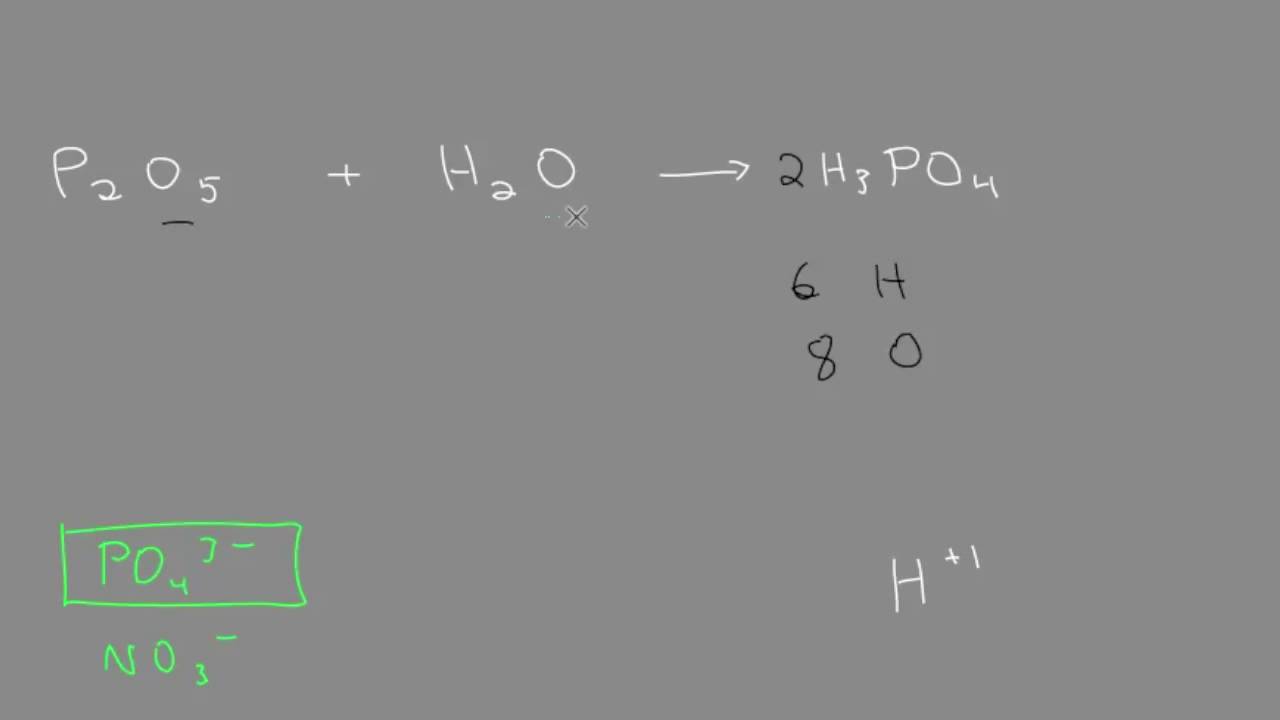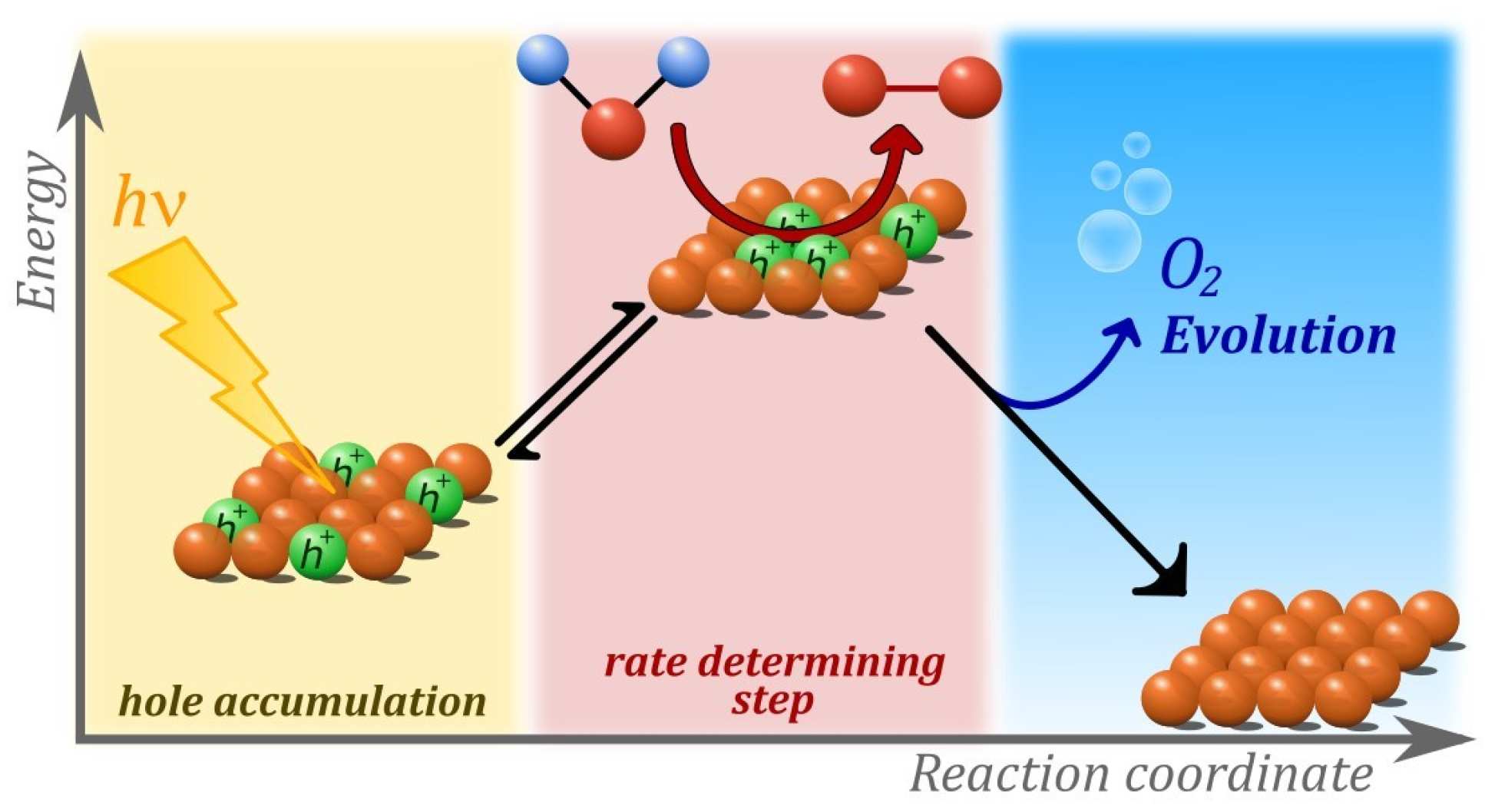
It takes three to make oxygen from water on metal oxides under sunlight | Imperial News | Imperial College London

Recent developments in the use of metal oxides for photocatalytic degradation of pharmaceutical pollutants in water—a review - ScienceDirect
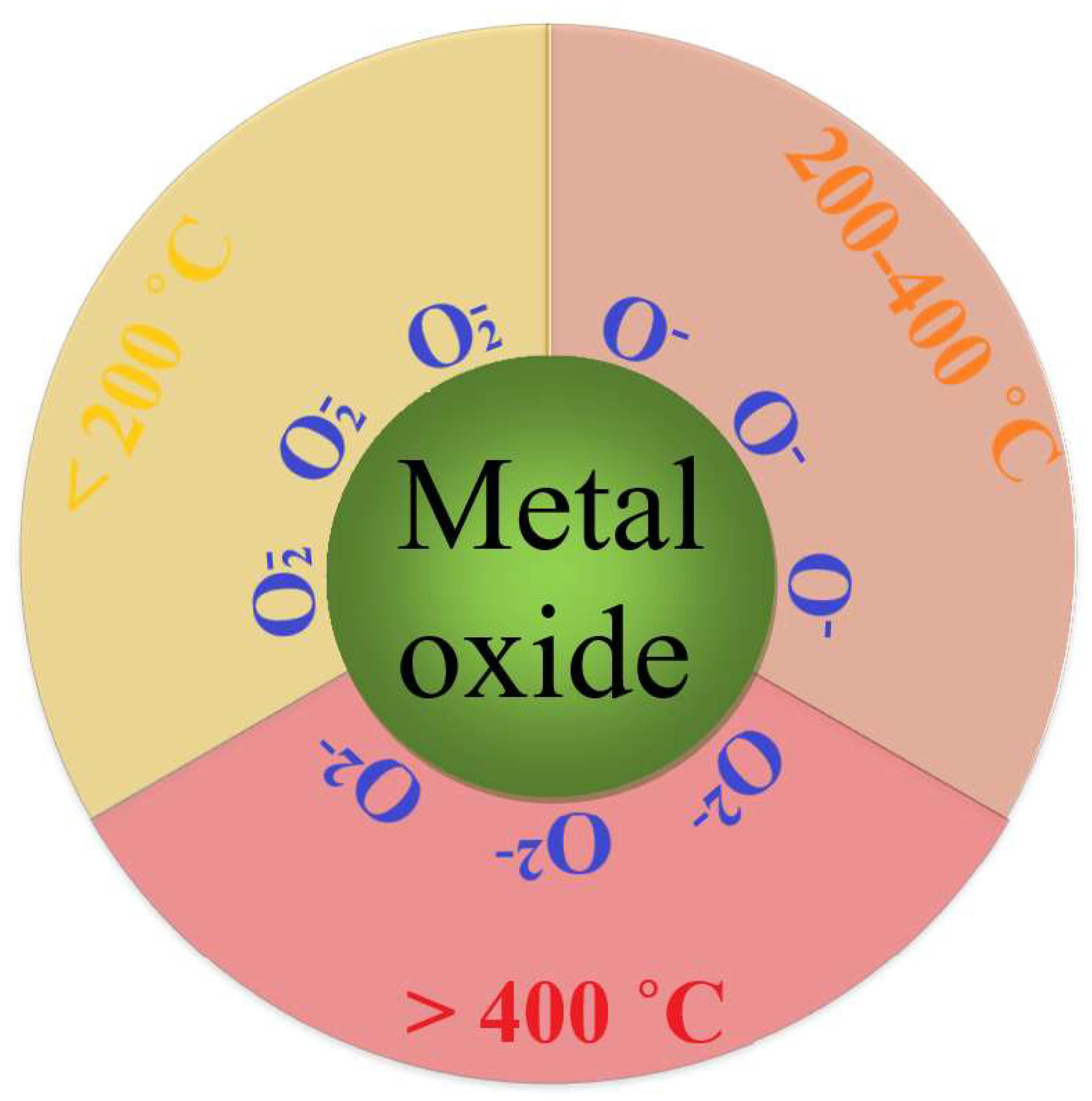
Chemosensors | Free Full-Text | Effect of Nanoparticle Interaction on Structural, Conducting and Sensing Properties of Mixed Metal Oxides

Thin water films covering oxide nanomaterials: Stability issues and influences on materials processing | Journal of Materials Research | Cambridge Core
Complete the reaction. Metal oxide + Dilute acid → SaIt + Water - Sarthaks eConnect | Largest Online Education Community

Metal + water give metal oxide + hydrogen Metal oxide + water give metal hydroxide Then what non metal + - Science - Metals and Non-metals - 13879007 | Meritnation.com

Towards a Molecular Level Understanding of the Multi-Electron Catalysis of Water Oxidation on Metal Oxide Surfaces | SpringerLink
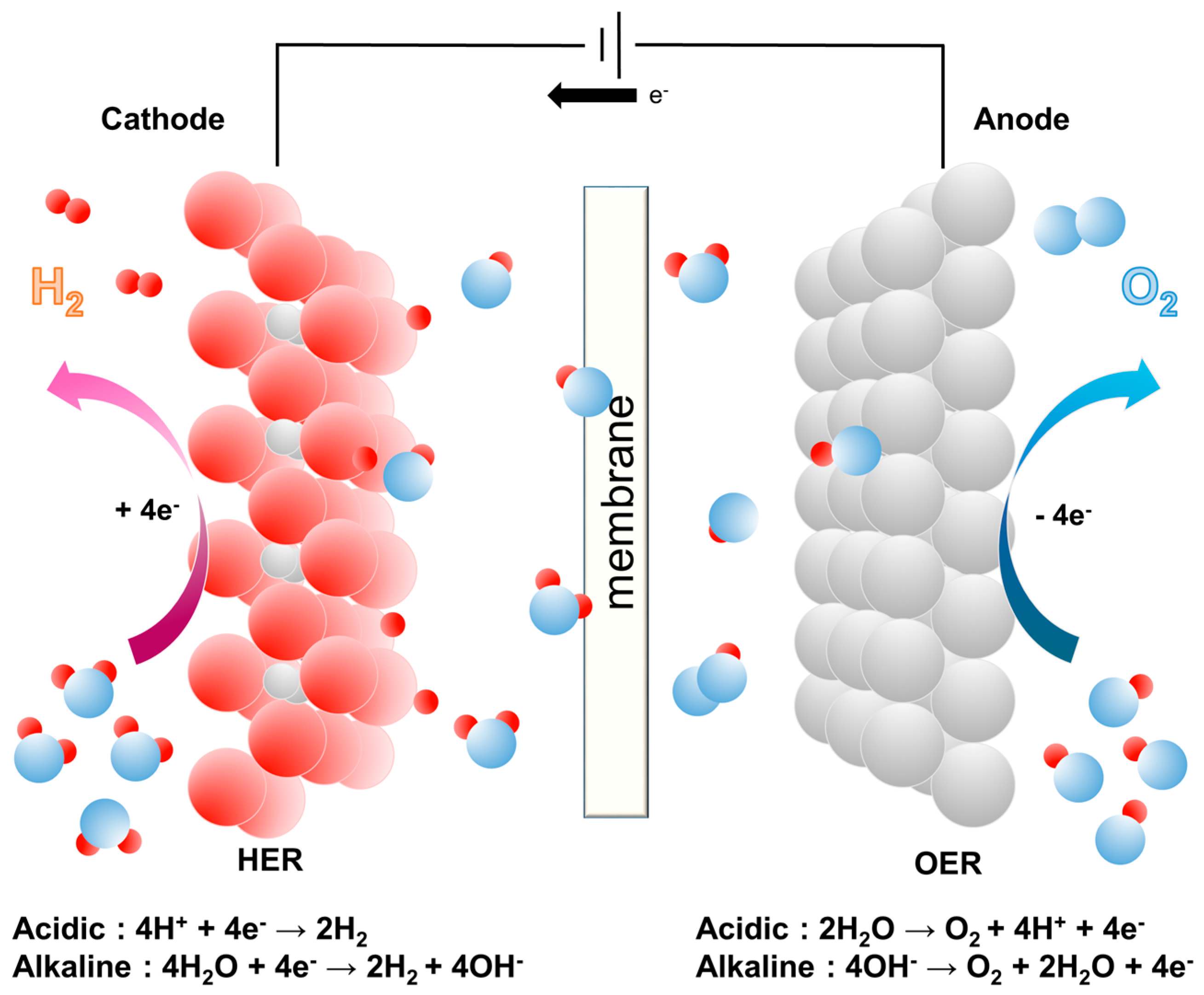
Materials | Free Full-Text | Multicomponent Metal Oxide- and Metal Hydroxide-Based Electrocatalysts for Alkaline Water Splitting

1) Metals react with water and produce a metal oxide and hydrogen gas. Metal oxides that are soluble in water dissolve in it to further form metal hydroxide. But all metals do